Celecoxib Synergistically Enhances MLN4924-Induced Cytotoxicity and EMT Inhibition Via AKT and ERK Pathways in Human Urothelial Carcinoma - Shida Xiong, Wei Huang, Xiaoqiang Liu, Qian Chen, Yi Ding, Haoxuan Huang, Ru Zhang,
Neddylation inhibitor MLN4924 suppresses growth and migration of human gastric cancer cells | Scientific Reports

Blockage of neddylation modification stimulates tumor sphere formation in vitro and stem cell differentiation and wound healing in vivo | PNAS
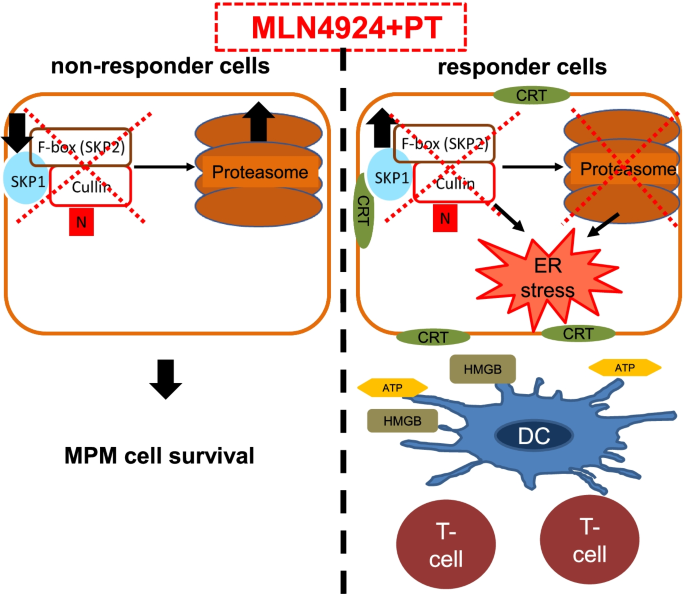
SKP2 drives the sensitivity to neddylation inhibitors and cisplatin in malignant pleural mesothelioma | Journal of Experimental & Clinical Cancer Research | Full Text
Inhibition of neddylation modification by MLN4924 sensitizes hepatocellular carcinoma cells to sorafenib
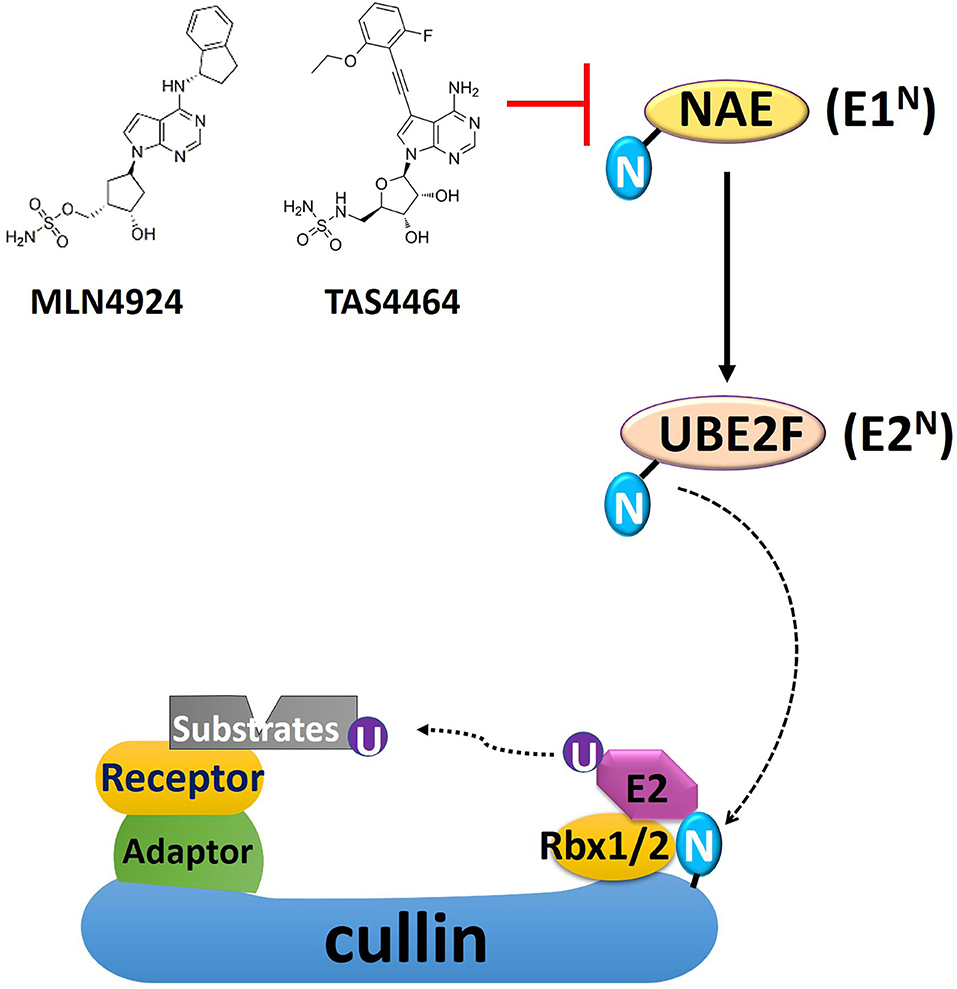
Frontiers | Targeting Cullin-RING E3 Ligases for Radiosensitization: From NEDDylation Inhibition to PROTACs

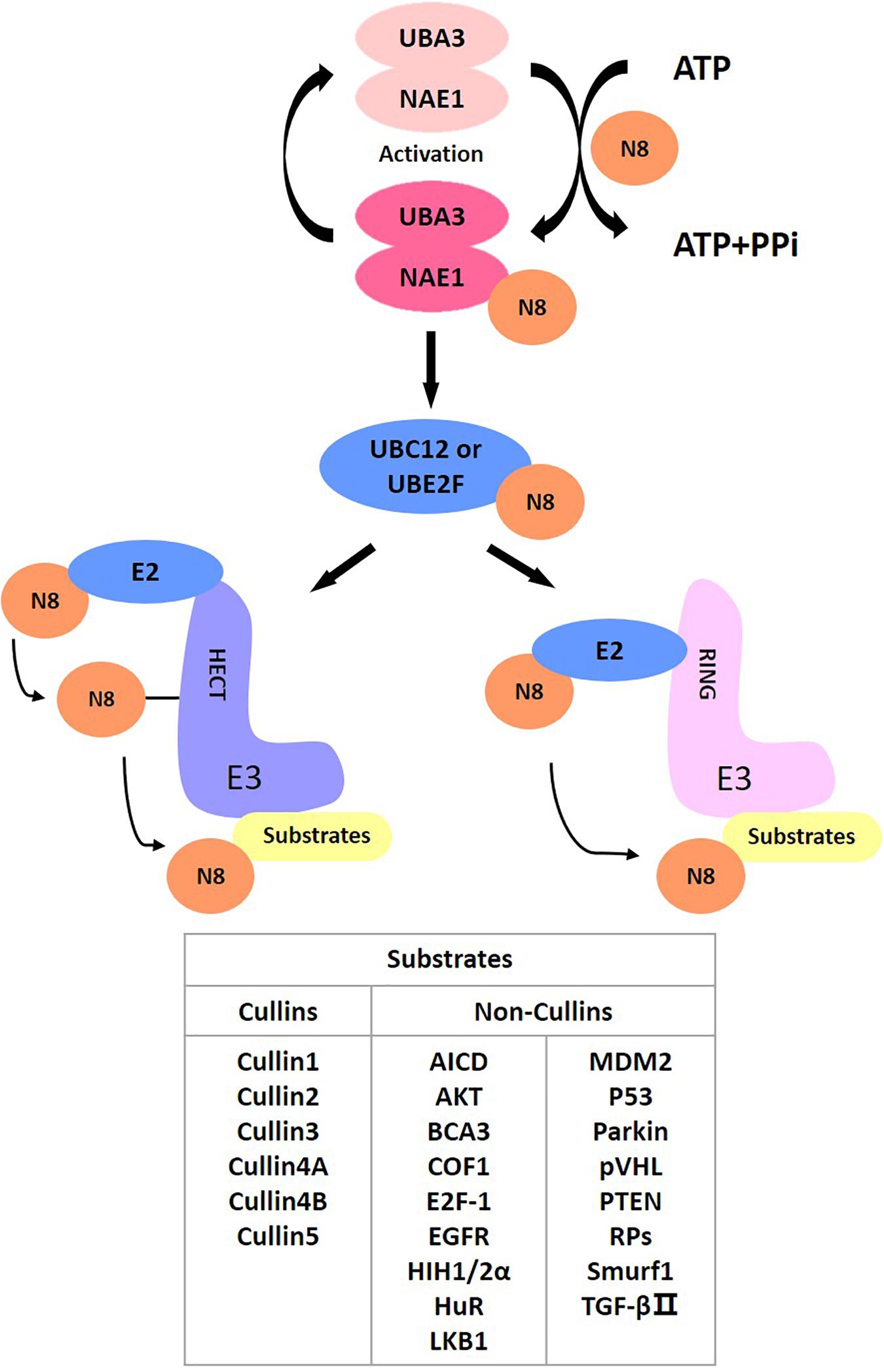
Targeting Neddylation Pathways to Inactivate Cullin-RING Ligases for Anticancer Therapy | Antioxidants & Redox Signaling
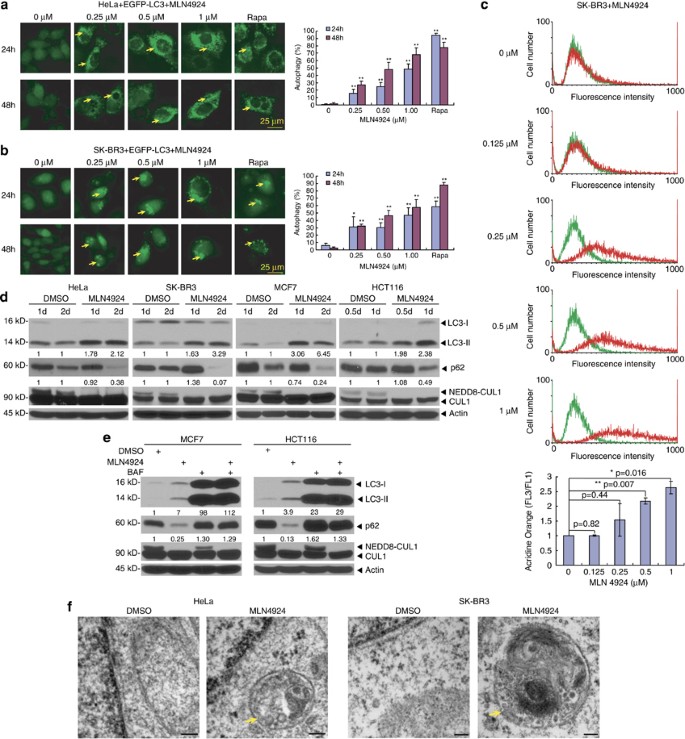
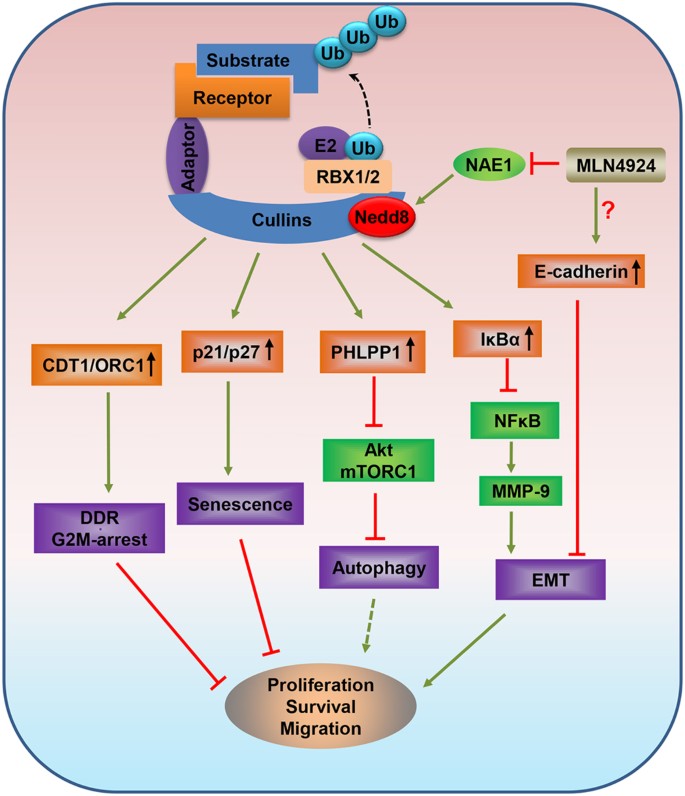
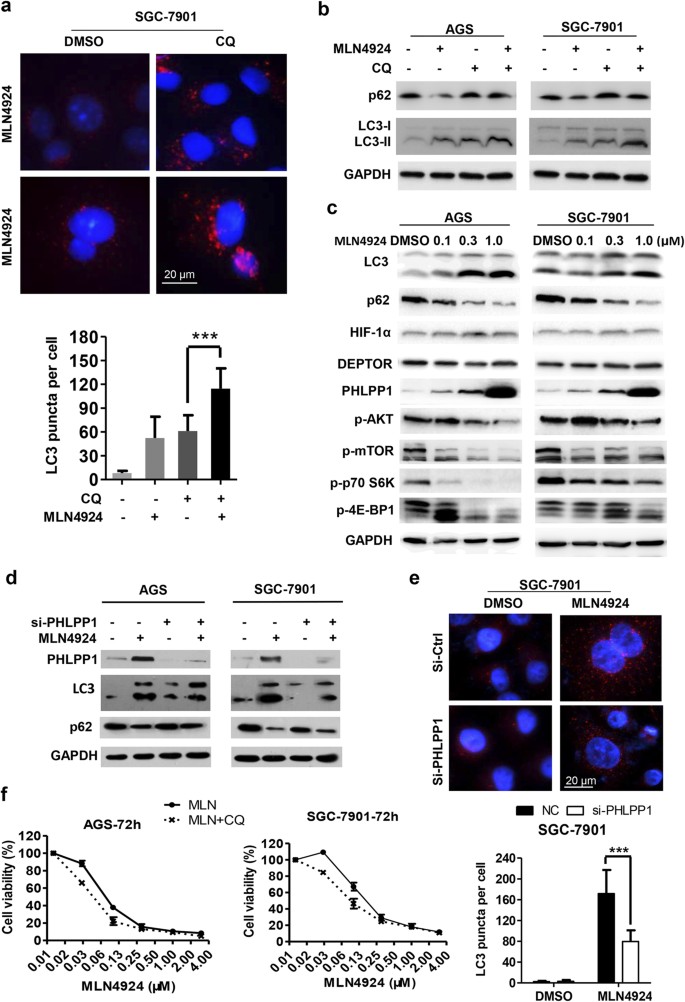
Targeting Cullin-RING ligases by MLN4924 induces autophagy via modulating the HIF1-REDD1-TSC1-mTORC1-DEPTOR axis | Cell Death & Disease

Cardioprotective effect of MLN4924 on ameliorating autophagic flux impairment in myocardial ischemia-reperfusion injury by Sirt1 - ScienceDirect

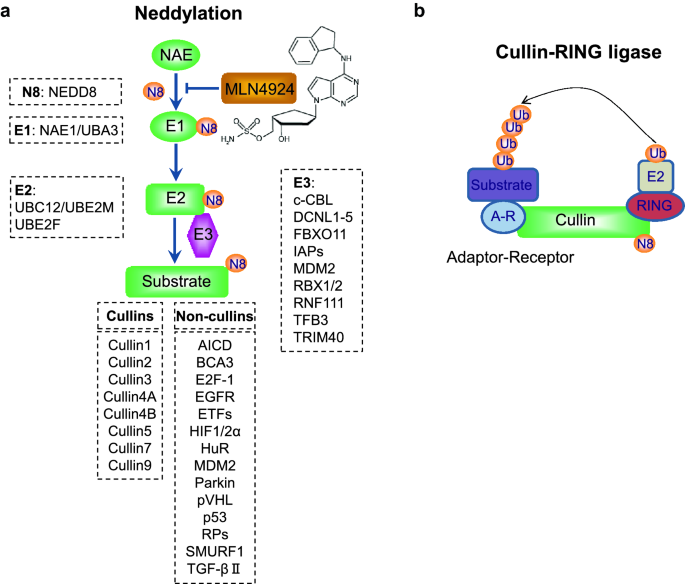
Neddylation Pathway as a Novel Anti-cancer Target: Mechanistic Investigation and Therapeutic Implication | Bentham Science

The MLN4924 inhibitor exerts a neuroprotective effect against oxidative stress injury via Nrf2 protein accumulation - ScienceDirect

Pevonedistat (MLN4924): mechanism of cell death induction and therapeutic potential in colorectal cancer | Cell Death Discovery
![PDF] The Nedd8-activating enzyme inhibitor MLN4924 induces autophagy and apoptosis to suppress liver cancer cell growth. | Semantic Scholar PDF] The Nedd8-activating enzyme inhibitor MLN4924 induces autophagy and apoptosis to suppress liver cancer cell growth. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/80cbd857543023682a105253c4bdb96a358339a4/4-Figure1-1.png)
PDF] The Nedd8-activating enzyme inhibitor MLN4924 induces autophagy and apoptosis to suppress liver cancer cell growth. | Semantic Scholar
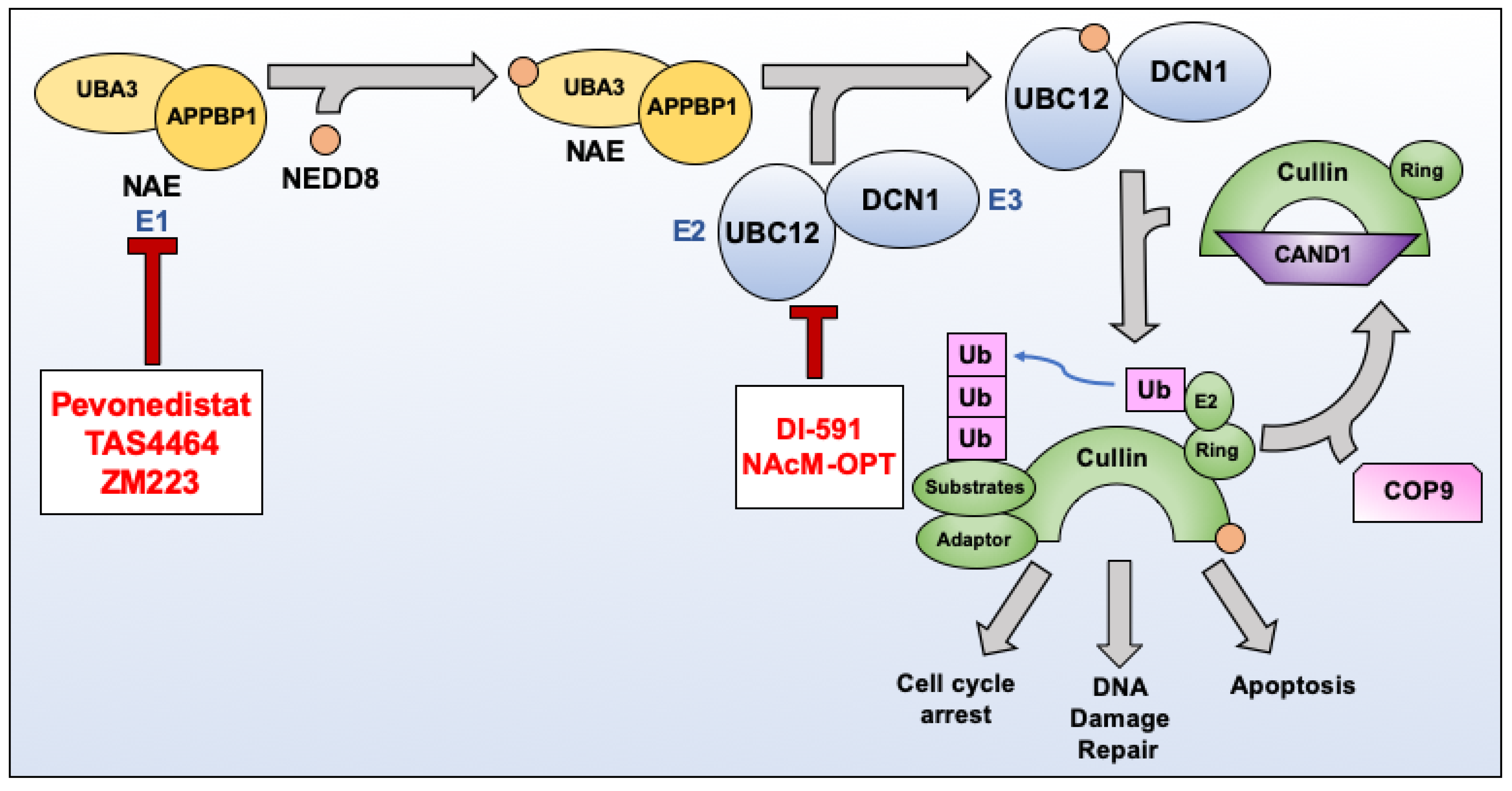
Cancers | Free Full-Text | Targeting NEDDylation as a Novel Approach to Improve the Treatment of Head and Neck Cancer
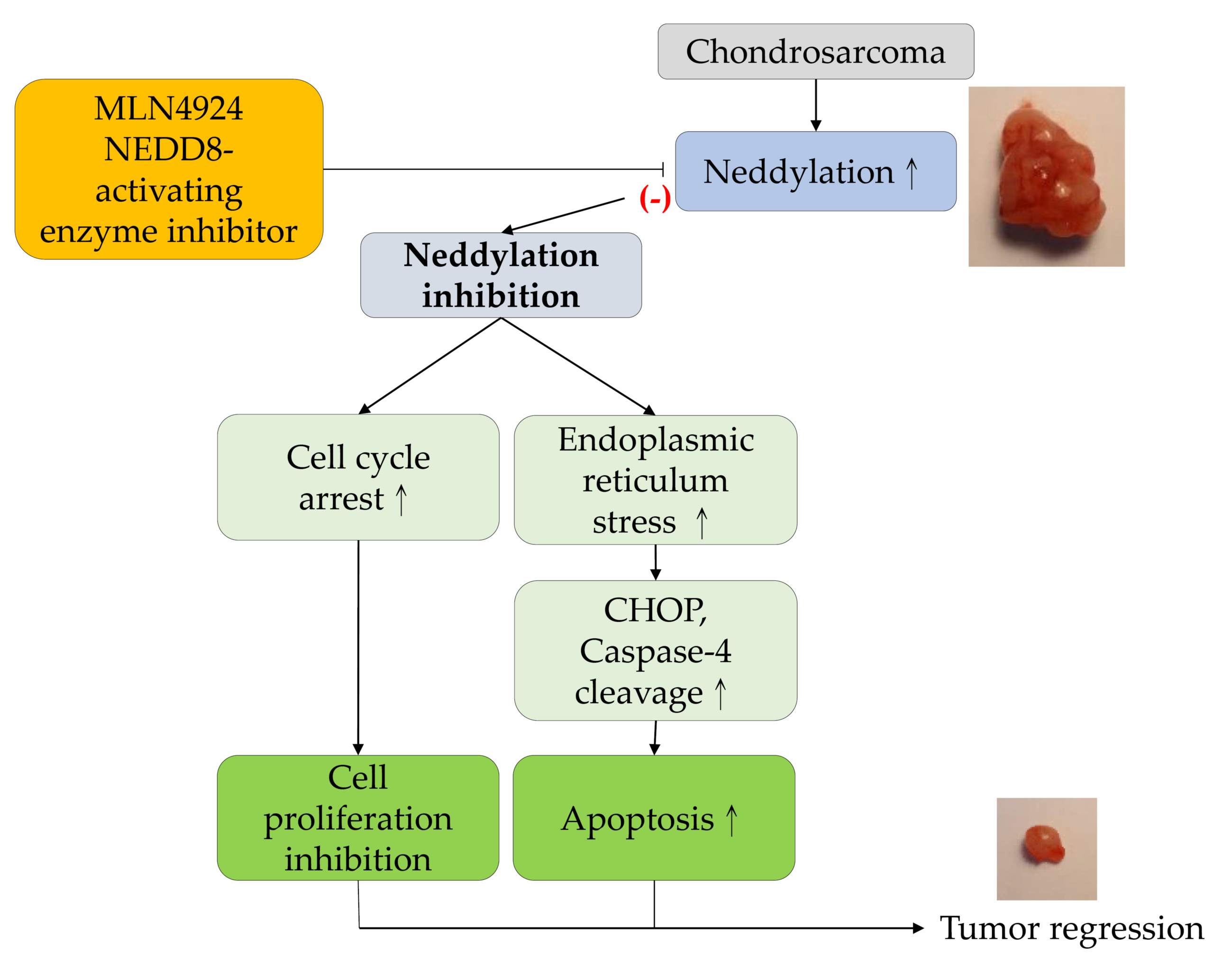
IJMS | Free Full-Text | MLN4924, a Protein Neddylation Inhibitor, Suppresses the Growth of Human Chondrosarcoma through Inhibiting Cell Proliferation and Inducing Endoplasmic Reticulum Stress-Related Apoptosis